Equipment;
Procedure
A cereal grain contains a store of starch within the endosperm. During germination the starch must be made soluble so that it can be transported to the embryo to support the growth of the seedling. The embryo is much smaller than the endosperm and is situated at the more pointed end of the grain. The developing embryo releases gibberellins that act on a layer of cells on the outside of the endosperm, stimulating these cells to release the starch-digesting enzyme amylase. In this activity you will remove the embryo and investigate the effect of different concentrations of gibberellin on the production of amylase.
The production of amylase will be assessed by using a starch agar assay. Cereal grains that have had the embryo removed are first soaked in gibberellic acid, then placed onto the starch agar plates and incubated. The agar plate is then flooded with iodine solution, which stains starch blue-black. The areas where starch has been digested will not stain. The size of the clear area around a cereal grain indicates the amount of amylase produced by the seed.
Planning
The basic procedure is outlined below but you will need to produce a plan before you start. This should provide detail and justify your decisions on how you will do the following.
Set the levels of the independent variable (gibberellin concentration). We will use a range of 6 different concentrations from 0 ugdm-3 to 500 ugdm-3. We are using intervals of 100 umgdm-3. 0cugdm-3 is our control variable to allow us yo compare what effect having no gibberellin has on an endosperm
Measure the dependent variable (size of the clear area).
Finally we need to ensure that we control any other factors that we are not measuring. This means that gibberellin concentration is the only variable that we are measuring making our experiment valid.
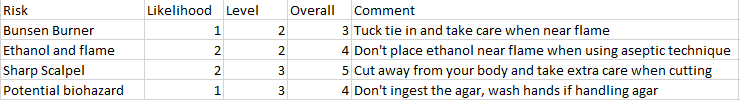
It is important that a good sterile technique is used throughout this investigation to prevent the growth of microorganisms that could present a biohazard.
Day 1
1. Make up your gibberellin solutions as planned. Only small volumes (a few cm3) of each are required. Place each solution in a small labelled sample bottle.
2. Collect the number of seeds that you require and pull any husks off the grains so the shape can be seen clearly.
3. Cut each seed across the line X-Y (see fig A) so that one half contains the embryo and the other the endosperm. Keep the two halves separate and discard the seed halves that contain the embryo.
4. Sterilise the remaining endosperm halves of the seeds by placing them in 3% sodium hypochlorite solution for 5 minutes.
5. Wash the seeds thoroughly, but quickly. through have changes of sterile water, draining carefully through muslin each time, until there is no smell of chlorine. Drain fully.
- gibberellic acid 1 gdm'3 stock solution
- 3% sodium hypochlorite bleach
- distilled water
- muslin
- Petri dishes with starch agar
- small beaker
- adhesive tape
- scalpel
- iodine in potassium iodide solution
- forceps
- volumetric glassware and measuring cylinders as required for dilutions of gibberellin
- small sterile containers for soaking grains
- tile
- marker pen
Procedure
A cereal grain contains a store of starch within the endosperm. During germination the starch must be made soluble so that it can be transported to the embryo to support the growth of the seedling. The embryo is much smaller than the endosperm and is situated at the more pointed end of the grain. The developing embryo releases gibberellins that act on a layer of cells on the outside of the endosperm, stimulating these cells to release the starch-digesting enzyme amylase. In this activity you will remove the embryo and investigate the effect of different concentrations of gibberellin on the production of amylase.
The production of amylase will be assessed by using a starch agar assay. Cereal grains that have had the embryo removed are first soaked in gibberellic acid, then placed onto the starch agar plates and incubated. The agar plate is then flooded with iodine solution, which stains starch blue-black. The areas where starch has been digested will not stain. The size of the clear area around a cereal grain indicates the amount of amylase produced by the seed.
Planning
The basic procedure is outlined below but you will need to produce a plan before you start. This should provide detail and justify your decisions on how you will do the following.
Set the levels of the independent variable (gibberellin concentration). We will use a range of 6 different concentrations from 0 ugdm-3 to 500 ugdm-3. We are using intervals of 100 umgdm-3. 0cugdm-3 is our control variable to allow us yo compare what effect having no gibberellin has on an endosperm
Measure the dependent variable (size of the clear area).
Finally we need to ensure that we control any other factors that we are not measuring. This means that gibberellin concentration is the only variable that we are measuring making our experiment valid.
It is important that a good sterile technique is used throughout this investigation to prevent the growth of microorganisms that could present a biohazard.
Day 1
1. Make up your gibberellin solutions as planned. Only small volumes (a few cm3) of each are required. Place each solution in a small labelled sample bottle.
2. Collect the number of seeds that you require and pull any husks off the grains so the shape can be seen clearly.
3. Cut each seed across the line X-Y (see fig A) so that one half contains the embryo and the other the endosperm. Keep the two halves separate and discard the seed halves that contain the embryo.
4. Sterilise the remaining endosperm halves of the seeds by placing them in 3% sodium hypochlorite solution for 5 minutes.
5. Wash the seeds thoroughly, but quickly. through have changes of sterile water, draining carefully through muslin each time, until there is no smell of chlorine. Drain fully.
 RSS Feed
RSS Feed
