Method:
1. Using aseptic technique, add 0.025 g dried yeast to 100 cm3 sterile distilled water at 30°C. Shake gently to ensure that the cells are evenly distributed and suspended.
2.Using aseptic technique, dilute 100 times by adding 1 cm3 to 99 cm3 sterile broth in a flask. This should give a startingconcentration of 0.0025 g/l for your growth curve.
3.Using sterile medium as the reference, calibrate the colorimeter (i.e. set it to zero).
4.Shake the flask containing the yeast culture gently to distribute the cells evenly. Using aseptic technique, withdraw a 1 cm3sample and add to 9cm3 of water.
5.Measure the absorbance of the sample you have just withdrawn. Record date, time and absorbance in the table.
6.Incubate at 30°C.
7.Repeat Instructions 5 - 8 three times per day for the next three days (early morning, lunch-time and late afternoon if possible). If it is not possible to measure the absorbance at the time of taking the sample, place it in a sterile container, label with initials, date and time and refrigerate until convenient to do so, preferably within 24 hours. Make sure that the yeast is fully suspended before reading the absorbance.
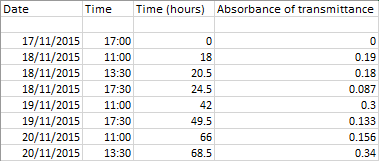
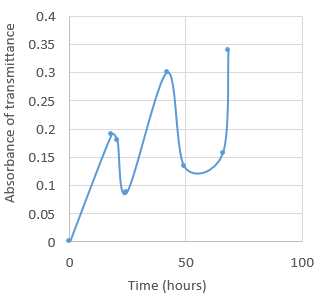
Results
1. Using aseptic technique, add 0.025 g dried yeast to 100 cm3 sterile distilled water at 30°C. Shake gently to ensure that the cells are evenly distributed and suspended.
2.Using aseptic technique, dilute 100 times by adding 1 cm3 to 99 cm3 sterile broth in a flask. This should give a startingconcentration of 0.0025 g/l for your growth curve.
3.Using sterile medium as the reference, calibrate the colorimeter (i.e. set it to zero).
4.Shake the flask containing the yeast culture gently to distribute the cells evenly. Using aseptic technique, withdraw a 1 cm3sample and add to 9cm3 of water.
5.Measure the absorbance of the sample you have just withdrawn. Record date, time and absorbance in the table.
6.Incubate at 30°C.
7.Repeat Instructions 5 - 8 three times per day for the next three days (early morning, lunch-time and late afternoon if possible). If it is not possible to measure the absorbance at the time of taking the sample, place it in a sterile container, label with initials, date and time and refrigerate until convenient to do so, preferably within 24 hours. Make sure that the yeast is fully suspended before reading the absorbance.
Results
Evaluation
The results we obtained do not show the expected results, although there is a general increase in the population of yeast shown by the increase in absorbance. There are a few inaccuracies in our method that may have effected the results. Half of our results were recorded as absorbance (a logarithmic scale from 0 to infinity usually kept between 0-1) and the other half as transmittance(a linear scale 0-100%). There were also negative values recorded that could not be converted to absorbance through the 2-log(T%)=absorbance, and therefore we could not get an accurate result. To improve this, we should have used another sample of the broth and evaluated the results of the new sample with the used one, if the negative results persisted we should use a different colorimeter.






 RSS Feed
RSS Feed
