Water is the second most common molecule in the universe, however it is also one of the most complex. Despite 330 million cubic miles of water surrounding on Earth, scientists are still perplexed by some of the properties shown by it. So what is so special about water?
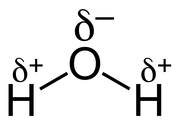
Water consists of only 3 atoms, 2 hydrogen bonded covalently to 1 oxygen atom. This structure creates a polar molecule, as the oxygen nucleus attracts the hydrogen electrons, thus creating a slightly positive charge to form by the hydrogen. The opposite occurs with the oxygen creating a slightly negative charge that end. This allows hydrogen bonds to form between different water molecules, which allows for it to display rather unique properties;
Most substances dissolve in water- the polarity of water allows it to pull apart ionic compounds, dissociation, this allows for ionic compounds that would otherwise need a large amount of energy to become a liquid, to instead be used as a solution. This property allows water to be an excellent transport medium, allowing plants to transfer essential ions easily dissolved in water.
Water is cohesive. The hydrogen bonds between the molecules mean that they stick together easily, this allows for water to travel up the plant in the xylem as it sticks to the lignin impregnated walls.
Water is also adhesive, they are attracted to different molecules. This is important in surface tension and plant transport systems.
Water has a high specific heat capacity, meaning it is slow to absorb a release heat. The hydrogen bonds between the molecules, result in a lot of energy being needed to separate them when heating. This allows for heating systems to work in homes as the heat is released slowly by radiators allowing the room to warm up. It also results in large bodies of water not changing much in temperature throughout the year, this allows the UK to have relatively nice weather compared to places on the same latitude.
Solid water(ice) floats on liquid water- a unique property due to the distance in hydrogen bonds being larger in ice and shorter in water. This property allowed life to thrive on the beds of lakes. A similar property which results in water being most dense at 4 degrees C, due to the spacing and strength of hydrogen bonds.
Water as a high surface tension, due to the attraction between the hydrogen molecules and the water below being greater then the attraction with the air above. This creates a thick layer of surface tension which allows for plant transport systems to work and for insects to skate across water.
The Mpemba Effect- perhaps the most strangest property which scientists have not figured out the true answer to yet. Water freezes faster from a higher temperature than a lower one, despite the temperature difference being greater freezing water from boiling takes a shorter time. Scientists have yet to discover the reason behind this phenomenon. However I personally back the idea that the hydrogen bonds have been stretched in boiling water and therefore do not have to be stretched again when water freezes.
Water in itself is an outstanding molecule, without it nothing would thrive on Earth.
Water consists of only 3 atoms, 2 hydrogen bonded covalently to 1 oxygen atom. This structure creates a polar molecule, as the oxygen nucleus attracts the hydrogen electrons, thus creating a slightly positive charge to form by the hydrogen. The opposite occurs with the oxygen creating a slightly negative charge that end. This allows hydrogen bonds to form between different water molecules, which allows for it to display rather unique properties;
Most substances dissolve in water- the polarity of water allows it to pull apart ionic compounds, dissociation, this allows for ionic compounds that would otherwise need a large amount of energy to become a liquid, to instead be used as a solution. This property allows water to be an excellent transport medium, allowing plants to transfer essential ions easily dissolved in water.
Water is cohesive. The hydrogen bonds between the molecules mean that they stick together easily, this allows for water to travel up the plant in the xylem as it sticks to the lignin impregnated walls.
Water is also adhesive, they are attracted to different molecules. This is important in surface tension and plant transport systems.
Water has a high specific heat capacity, meaning it is slow to absorb a release heat. The hydrogen bonds between the molecules, result in a lot of energy being needed to separate them when heating. This allows for heating systems to work in homes as the heat is released slowly by radiators allowing the room to warm up. It also results in large bodies of water not changing much in temperature throughout the year, this allows the UK to have relatively nice weather compared to places on the same latitude.
Solid water(ice) floats on liquid water- a unique property due to the distance in hydrogen bonds being larger in ice and shorter in water. This property allowed life to thrive on the beds of lakes. A similar property which results in water being most dense at 4 degrees C, due to the spacing and strength of hydrogen bonds.
Water as a high surface tension, due to the attraction between the hydrogen molecules and the water below being greater then the attraction with the air above. This creates a thick layer of surface tension which allows for plant transport systems to work and for insects to skate across water.
The Mpemba Effect- perhaps the most strangest property which scientists have not figured out the true answer to yet. Water freezes faster from a higher temperature than a lower one, despite the temperature difference being greater freezing water from boiling takes a shorter time. Scientists have yet to discover the reason behind this phenomenon. However I personally back the idea that the hydrogen bonds have been stretched in boiling water and therefore do not have to be stretched again when water freezes.
Water in itself is an outstanding molecule, without it nothing would thrive on Earth.

 RSS Feed
RSS Feed
